Anesthesiologist Tech Gadgets: Top 5 Picks Reviewed
Medical professionals demand precision, reliability, and cutting-edge technology to deliver exceptional patient care. Anesthesiologists, in particular, rely on sophisticated gadgets and devices to monitor vital signs, manage medications, and maintain patient safety during surgical procedures. The intersection of healthcare and technology continues to evolve, offering professionals access to innovative tools that enhance their capabilities in the operating room.
Whether you’re an experienced anesthesiologist seeking to upgrade your toolkit or a resident exploring the latest advancements in anesthesia technology, understanding which gadgets deliver genuine value is essential. This comprehensive review examines five outstanding tech solutions specifically designed for anesthesia professionals, analyzing their features, performance, and real-world applications in clinical settings.
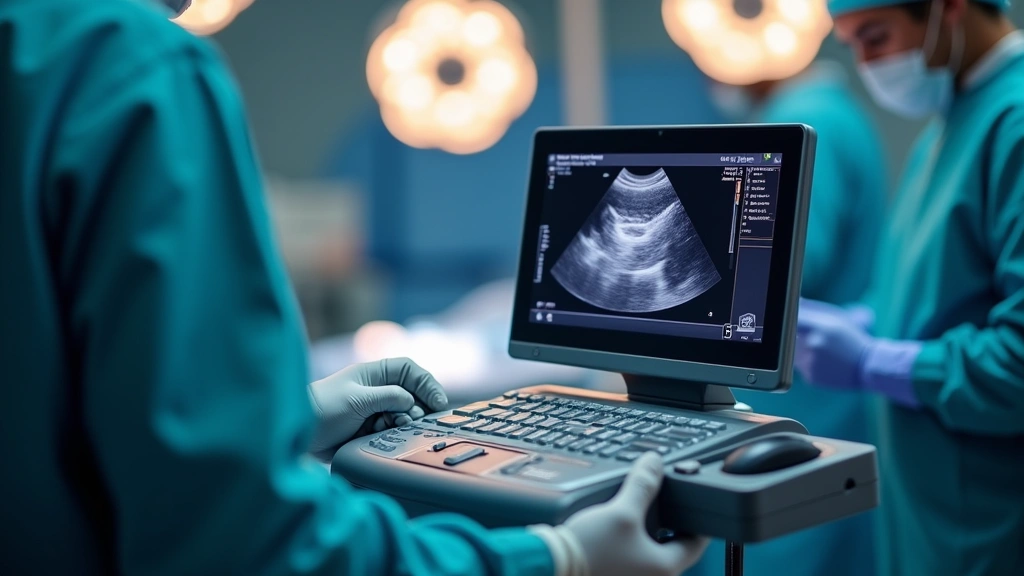
Portable Ultrasound Systems for Nerve Blocks
Portable ultrasound technology has revolutionized regional anesthesia techniques, enabling precise nerve localization and medication delivery. Modern handheld ultrasound systems designed for anesthesiologist tech applications combine exceptional image quality with ergonomic designs suitable for operating room environments.
The latest generation of portable ultrasound devices features high-frequency probes delivering superior resolution for vascular access and nerve identification. These systems typically weigh between 1.5 to 3 pounds, making them easily transportable between operating rooms and procedure areas. Advanced image processing algorithms enhance contrast and clarity, allowing clinicians to distinguish anatomical structures with remarkable precision even in challenging patient populations.
Key specifications include real-time imaging at 60+ frames per second, color Doppler capabilities for vascular assessment, and battery life extending 8-12 hours on a single charge. Many professional-grade models offer touchscreen interfaces with customizable presets for common regional anesthesia procedures. The integration of AI-assisted measurements helps calculate optimal needle trajectories and medication volumes.
Clinical performance data demonstrates that portable ultrasound systems reduce procedure time by 25-40% compared to landmark-based techniques, while simultaneously improving success rates on first attempt. The ability to visualize needle advancement in real-time significantly enhances safety outcomes and patient satisfaction. Integration with hospital networks allows for seamless documentation and image archiving directly to electronic health records.
Investment in quality portable ultrasound equipment represents a significant but worthwhile expense, with systems ranging from $15,000 to $45,000 depending on specifications and probe options. Consider systems with expandable probe libraries to accommodate evolving clinical needs and future technological upgrades.
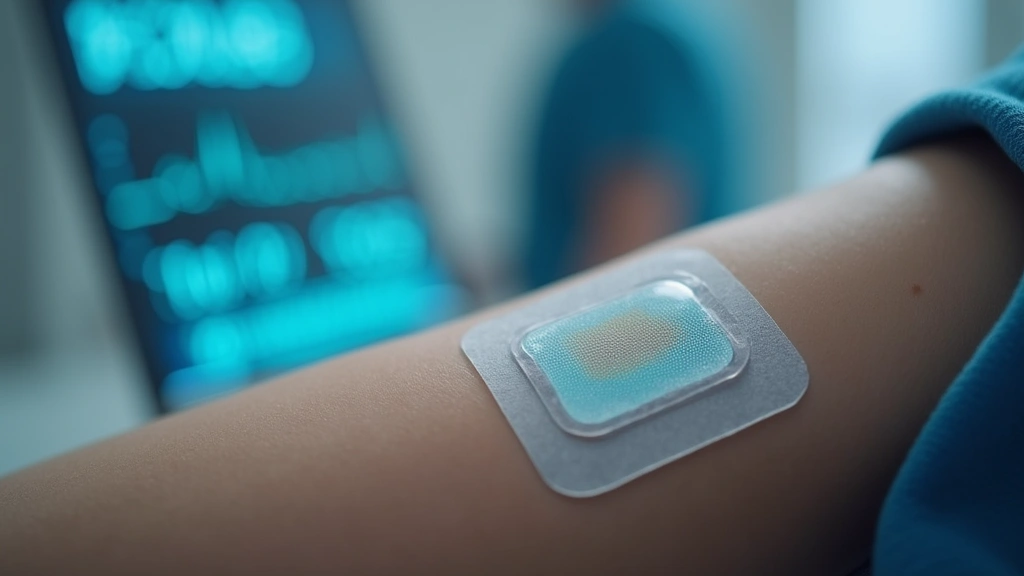
Advanced Patient Monitoring Wearables
Continuous patient monitoring throughout the perioperative period demands sophisticated wearable technology that provides real-time data without impeding surgical access. Contemporary wearable monitoring devices designed for anesthesiologists integrate multiple sensors capturing heart rate variability, oxygen saturation, core temperature, and non-invasive blood pressure readings simultaneously.
These advanced wearables utilize flexible electrode arrays and biocompatible adhesive formulations, allowing secure placement on patient skin for extended monitoring periods without irritation or displacement. Wireless transmission protocols ensure data flows continuously to central monitoring stations with minimal latency—typically under 500 milliseconds—critical for detecting rapid physiological changes during anesthesia.
Premium wearable systems incorporate machine learning algorithms that establish individualized baseline parameters for each patient, enabling predictive alerting before critical thresholds are reached. This proactive approach allows anesthesiologists to intervene earlier, potentially preventing complications before they develop into serious events. The technology analyzes complex relationships between multiple vital parameters simultaneously, identifying subtle patterns humans might miss.
Battery longevity represents a crucial specification, with leading models maintaining continuous operation for 24-48 hours. Charging interfaces utilize standard USB-C connectors, facilitating rapid turnaround between patients. Data storage capacity typically accommodates 72+ hours of continuous monitoring at maximum sampling rates, providing comprehensive records for quality assurance and research purposes.
Integration capabilities with existing hospital infrastructure vary significantly. The most versatile systems support HL7 and FHIR data standards, enabling seamless communication with electronic health records and clinical decision support systems. Some manufacturers offer compatible tablet applications for remote monitoring from multiple locations within the hospital.
Accuracy specifications for wearable monitoring typically match or exceed traditional bedside monitors in most parameters. However, motion artifact during surgical procedures remains a consideration—leading devices employ adaptive filtering algorithms to distinguish true physiological signals from movement-related noise. Clinical validation studies show accuracy rates exceeding 95% for heart rate and oxygen saturation measurements even during active surgery.
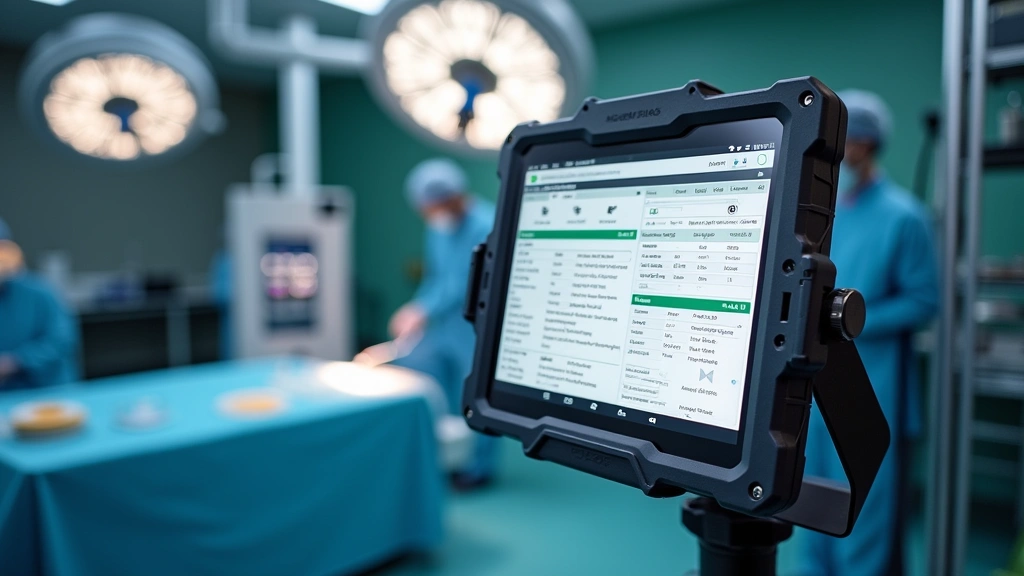
Mobile Anesthesia Documentation Tablets
Efficient anesthesia record documentation directly impacts patient safety, billing accuracy, and compliance with regulatory requirements. Specialized tablets designed specifically for operating room use streamline documentation workflows while maintaining HIPAA compliance and data security standards.
Purpose-built anesthesia documentation tablets feature ruggedized construction designed to withstand repeated sterilization cycles, accidental drops, and exposure to surgical environments. Antimicrobial coating on touchscreens prevents bacterial colonization and cross-contamination between patients. The devices typically utilize gorilla glass or equivalent protection rated for operating room durability.
Processing power in modern anesthesia tablets must support simultaneous operation of multiple applications without lag—anesthesia record software, vital sign monitoring interfaces, pharmacy databases, and hospital network connectivity all operate concurrently. Minimum specifications should include quad-core processors operating at 2.5+ GHz, 6GB RAM, and high-speed storage with 256GB capacity.
Display technology matters significantly for operating room visibility. Anti-glare screens with brightness levels exceeding 500 nits ensure readability under surgical lighting conditions. Some premium models incorporate polarization filters to reduce reflections and improve contrast when viewed from multiple angles—essential when multiple team members need to reference information simultaneously.
Integration with electronic health records systems represents perhaps the most critical functionality. Leading anesthesia documentation tablets synchronize automatically with hospital networks, providing real-time access to patient history, medication allergies, previous anesthesia records, and current vital signs. The systems employ sophisticated data validation rules that flag potential medication errors, drug interactions, or contraindications before administration.
Battery performance in tablets designed for anesthesia must support 8-10 hour operating room shifts on a single charge, with rapid charging capabilities enabling 30-minute turnaround between cases. Wireless connectivity options should include both Wi-Fi 6 (802.11ax) and cellular backup to maintain connectivity even if hospital networks experience disruptions.
Voice recognition features increasingly integrated into anesthesia documentation tablets allow hands-free input during critical moments when manual data entry isn’t feasible. These systems employ specialized medical vocabulary training to accurately transcribe anesthesia-specific terminology and dosing information, reducing documentation errors compared to manual entry.
Smart Infusion Pump Controllers
Medication administration accuracy directly determines anesthesia quality and patient safety outcomes. Contemporary smart infusion pump controllers combine precise flow rate management with sophisticated software algorithms that prevent common medication errors.
These devices function as intelligent intermediaries between traditional infusion pumps and hospital pharmacy systems, creating closed-loop medication administration workflows. Smart controllers validate every medication bolus or infusion rate against patient-specific parameters—weight-based calculations, drug interaction checks, renal function considerations, and institutional protocols—before allowing administration to proceed.
Technical specifications for leading smart infusion controllers include flow rate accuracy within ±2% across the full operational range, typically 0.1 mL/hour to 1200 mL/hour depending on pump model. Pressure monitoring sensors detect infiltration, occlusion, or line displacement, triggering immediate alarms and halting medication delivery to prevent patient harm.
Integration capabilities with electronic health records and pharmacy systems enable bidirectional data flow. The controller pulls patient-specific parameters and medication orders directly from hospital systems, eliminating manual entry errors. Simultaneously, it logs administration records automatically, creating audit trails that satisfy regulatory compliance requirements and support quality improvement initiatives.
Wireless connectivity in smart controllers must prioritize security without compromising responsiveness. Leading manufacturers employ military-grade encryption protocols (AES-256) protecting all data transmission over hospital networks. Authentication mechanisms typically utilize multi-factor verification, ensuring only authorized personnel can modify infusion parameters or override safety constraints.
Alarm management represents a sophisticated feature in premium controllers. Rather than generating excessive alerts that lead to alarm fatigue, intelligent systems prioritize notifications based on clinical significance and patient risk factors. Customizable alert thresholds allow individual institutions to tailor sensitivity levels to their specific protocols and patient populations.
Battery backup in smart infusion controllers maintains functionality for 4+ hours during power disruptions, ensuring continuous medication delivery despite facility-wide electrical failures. Automatic failover to backup power systems occurs seamlessly without interrupting infusions or requiring user intervention.
Wireless Vital Signs Monitoring Devices
Comprehensive vital signs monitoring throughout the perioperative period remains fundamental to anesthesia management. Wireless monitoring devices designed for modern operating rooms eliminate cumbersome cables while maintaining robust data transmission and clinical accuracy standards.
Contemporary wireless vital signs monitors utilize proprietary frequency-hopping protocols operating in the 2.4 GHz ISM band, providing reliable transmission in electromagnetically challenging operating room environments. Advanced error correction algorithms ensure data integrity even when multiple wireless devices operate simultaneously within the same facility.
Sensor technology in wireless monitors continues advancing, with newer devices incorporating non-invasive cardiac output monitoring, tissue oxygenation assessment, and neuromuscular function measurement alongside traditional parameters. These comprehensive metrics provide anesthesiologists with unprecedented insight into patient physiology, enabling more precise titration of anesthetic agents and faster recognition of physiological deterioration.
Display interfaces on wireless monitoring systems employ large, high-contrast numerical readouts visible from across the operating room. Trend graphs showing parameter evolution over time help clinicians identify gradual physiological changes that might be missed when reviewing only instantaneous values. Color-coded alert zones provide intuitive visual feedback regarding parameter normality—green for safe ranges, yellow for caution, red for critical values.
Data logging capabilities in wireless monitors typically store 24+ hours of continuous monitoring data at maximum resolution, enabling comprehensive review of patient physiology throughout hospitalization. Export functions facilitate transfer to electronic health records and research databases for quality improvement analysis and clinical studies.
Calibration requirements for wireless monitoring devices vary by sensor type. Most manufacturers design contemporary sensors for minimal calibration needs—many utilize factory calibration lasting the entire sensor lifespan. This reduces workflow interruptions and potential calibration errors compared to older devices requiring frequent manual calibration between patients.
Regulatory compliance for wireless medical devices includes FCC certification in North America, CE marking in Europe, and equivalent certifications in other markets. When evaluating wireless monitoring systems, verify that devices carry appropriate regulatory approvals for your jurisdiction and meet electromagnetic compatibility standards required for hospital environments.
The integration of wireless vital signs monitoring with advanced clinical decision support systems enables predictive analytics that identify patients at risk for perioperative complications before critical events occur. Machine learning algorithms trained on thousands of patient records can recognize subtle physiological patterns associated with adverse outcomes, providing early warning to the anesthesia team.
FAQ
What budget should anesthesiologists allocate for technology gadgets?
Investment requirements vary based on practice setting and clinical needs. Portable ultrasound systems typically represent the largest single investment ($15,000-$45,000), while tablets and wearable monitors range from $3,000-$15,000 each. Many institutions amortize these costs over multiple years, and some equipment qualifies for capital equipment tax deductions. Consider total cost of ownership including maintenance, software licenses, and replacement sensors when budgeting.
How do anesthesiologist tech gadgets integrate with existing hospital systems?
Modern devices employ standardized data formats (HL7, FHIR) enabling integration with electronic health records and hospital networks. Most manufacturers provide IT support for integration implementation. However, compatibility varies significantly—verify integration capabilities with your specific hospital systems before purchasing. Some institutions require security assessments and network validation before deploying new wireless devices.
What training is required to effectively use advanced anesthesia technology?
Most manufacturers provide comprehensive training programs ranging from 2-8 hours for basic operation to advanced features. Many offer online training modules accessible on-demand, supplemented by hands-on practice sessions. Professional organizations including the American Society of Anesthesiologists frequently offer continuing education programs covering new technologies. Consider allocating training time for the entire anesthesia team, including residents and nurse anesthetists.
How frequently do anesthesia tech gadgets require maintenance and calibration?
Maintenance schedules vary by device type. Ultrasound systems typically require annual professional servicing ($500-$1,500). Tablets need routine software updates and periodic screen replacements ($200-$500). Wearable sensors generally don’t require calibration but may need battery replacement annually. Infusion pump controllers usually need annual validation testing. Budget approximately 10-15% of equipment purchase price annually for maintenance and repairs.
What regulatory considerations apply to anesthesiologist tech purchases?
All medical devices must carry appropriate regulatory approvals for your jurisdiction (FDA approval in the United States, CE marking in Europe). Wireless devices require electromagnetic compatibility testing. Some devices fall under HIPAA regulations regarding patient data protection. Before purchasing, verify that devices comply with relevant regulations and that your institution’s compliance team has approved deployment. Consider consulting with your hospital’s biomedical engineering department regarding compatibility with existing infrastructure.
How do I evaluate whether new technology will genuinely improve patient outcomes?
Request peer-reviewed clinical evidence supporting manufacturer claims. Look for independent validation studies published in anesthesia journals rather than relying solely on manufacturer data. Many professional organizations maintain technology assessment committees providing objective evaluations. Consider pilot programs where you can evaluate technology with a subset of cases before full institutional adoption. Involve your quality improvement team in tracking relevant metrics before and after implementation.
What cybersecurity measures protect patient data in wireless anesthesia devices?
Leading manufacturers employ military-grade encryption (AES-256) for data transmission and storage. Most devices require multi-factor authentication for access. Look for devices with automatic security updates, intrusion detection systems, and audit logging capabilities. Ensure devices comply with HIPAA security requirements and consider additional protections through your hospital’s cybersecurity infrastructure. Request security documentation and penetration testing results from manufacturers before deployment.